Inspire TMS Denver, a leading provider of advanced neuromodulation therapies, has announced the launch of its innovative ONE-D protocol, a Single-Day TMS treatment designed to deliver a full course of 20 sessions within one to two days. This new offering from Inspire TMS Denver represents a major advancement in accessibility for individuals seeking effective mental health care without the traditional six-to-nine-week commitment.
The introduction of Single-Day TMS addresses one of the most common barriers to treatment: time. Patients such as working professionals, parents, caregivers, and out-of-state travelers often struggle to commit to standard schedules. By condensing the full treatment course into a rapid timeframe, this One-Day TMS Denver solution opens new pathways for those previously unable to pursue care.

“Time is the single biggest reason patients don't start TMS,” said Dr. Samuel Clinch, board-certified psychiatrist and founder of Inspire TMS Denver. “ONE-D removes that barrier without cutting corners on the clinical work that makes treatment succeed.”
What sets this ONE-D TMS protocol apart is its emphasis on clinical precision. Unlike many clinics where technicians handle key steps, Dr. Samuel Clinch personally conducts motor-threshold mapping and coil positioning for every patient. This hands-on approach ensures accurate targeting and consistency across all sessions. It supports improved outcomes for individuals undergoing TMS therapy in Denver.
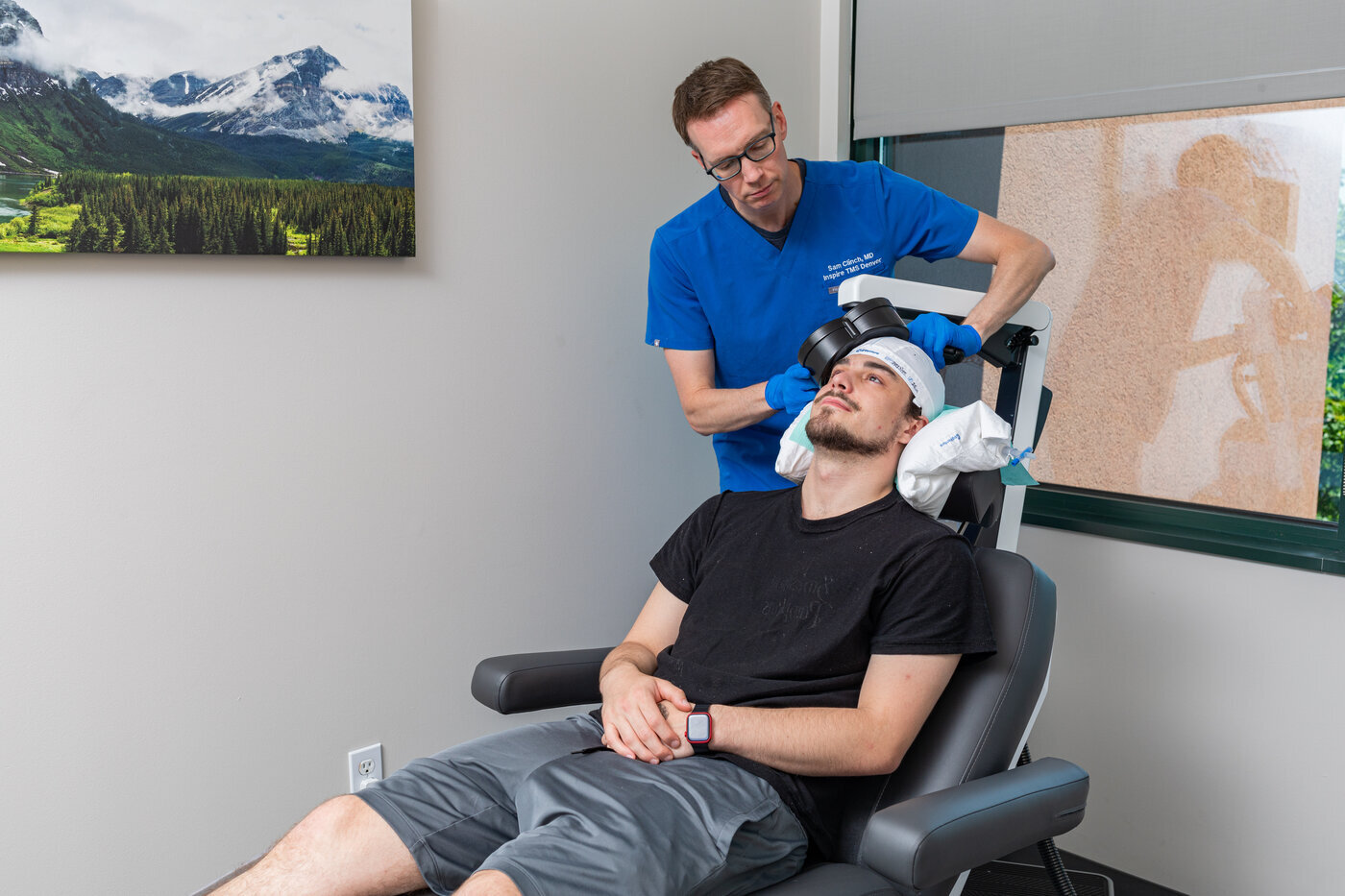
The clinic’s results reinforce the effectiveness of its protocols. Inspire TMS Denver reports response rates of 81% for depression, 73% for anxiety, 64% for OCD, and 76% for PTSD - figures that exceed the industry benchmark of 69% response and 36% remission reported in the NeuroStar patient registry. These outcomes are supported by the clinic’s use of advanced MagVenture targeted coil systems across all treatments, including standard, 5-day accelerated, and accelerated TMS Denver offerings.
Affordability is another key component of the ONE-D program. Inspire TMS Denver offers a flat self-pay rate of $3,500, which includes mapping, all 20 sessions, and follow-up care. This pricing is notably lower than the $7,000 or more often associated with similar accelerated protocols, making depression treatment in Denver more accessible to a broader population.

Located in Broomfield, Colorado, the clinic serves both the Denver metro area and patients traveling from across the country. For out-of-state visitors, Inspire TMS Denver provides logistical support, including hotel recommendations. This ensures a smooth and convenient treatment experience at its TMS Broomfield, CO facility. Since its founding, the clinic has delivered more than 15,000 treatments, building a strong track record of patient care and innovation.
Patients exploring alternatives to medication or seeking faster results can now consider the ONE-D protocol as a practical and effective option. By combining speed, precision, and affordability, Inspire TMS Denver continues to redefine what is possible in modern mental health care.
Those interested in learning more about this breakthrough single-day TMS option are encouraged to visit the website at https://www.inspiretmsdenver.com/ to explore treatment details and schedule a consultation.
About Inspire TMS Denver
Inspire TMS Denver is a Broomfield-based clinic specializing in advanced transcranial magnetic stimulation treatments for depression, anxiety, OCD, and PTSD. Led by Dr. Samuel Clinch, the clinic provides personalized care using evidence-based protocols and cutting-edge technology.
###
Media Contact
Inspire TMS Denver
340 E 1st Ave Suite 333, Broomfield, CO 80020
(720) 446-8675
https://www.inspiretmsdenver.com/
Disclaimer:
Any advice or guidelines revealed here are not even remotely a substitute for sound medical advice from a licensed healthcare provider. Make sure to consult with a professional physician before making any purchasing decision if you use medications or have concerns following the information and details shared above. Individual results may vary as the statements made regarding these products have not been evaluated by the Food and Drug Administration. The efficacy of these products has not been confirmed by FDA-approved research. These products are not intended to diagnose, treat, cure or prevent any disease.
*These statements have not been evaluated by the U.S. Food and Drug Administration. This product is not intended to cure or treat any disease.







